...
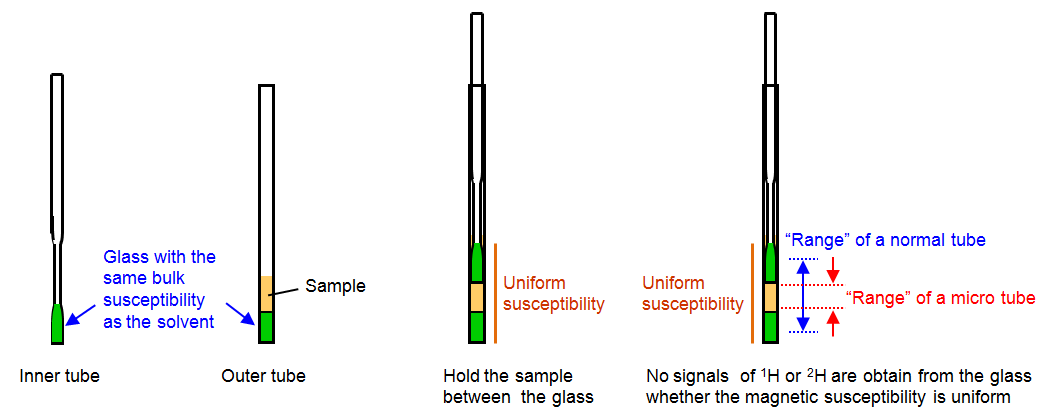
If you use a regular 5 mm NMR tube, a filling length of 40 mm is recommended, which corresponds to about 550 µl of sample in a 5 mm tube with walls 0.4 mm thick. If the volume is limited, 5 mm Shigemi (ie containing susceptibility-matched plugs), or 3 mm tubes (with slightly reduced sensitivity), can be used and will require less volume. Below is an illustration of the construction of a Shigemi tube:
Source: JEOL
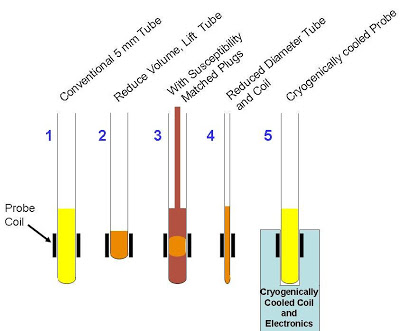
Summary of optimal volume and quatity quantity for different NMR tubes:
| Type of tube | Volume, µl | Quantity, nmol | |
|---|---|---|---|
| at 100 µM | at 500 µM | ||
| 5 mm | 550 | 55 | 275 |
| 5 mm Shigemi | 350 | 40 | 200 |
| 3 mm | 160 | 20 | 100 |
Practically, on the instruments of the Structural Biology Platform, at least 100 µM of sample is sufficient for simple checks (1D and 2D correlation experiments), whereas concentrations beyond 0.5 mM should be attained in order to have good SNR on multi-dimensional multidimensional experiments within acceptable experiment time. When the quantity and volume are limited, some options are available to obtain a good signal-to-noise ratio (SNR):
Source: UOttawa NMR facility blog
...
Salt (typically in the form of NaCl or KCl) is generally used in sample buffers in order to prevent non specific ionic interactions and favor proper folding of the sample. For NMR analysis, ionic strength with a salt concentrations concentration up to 500 mM is generally appropriate (below 150 mM being ideal). Higher salt concentrations complicate the tuning and matching of the probe and result in longer 90° pulse duration, which may need power adjustments exceeding maximal permitted levels for a given probe. This is particularly true for the cryoprobe.
...
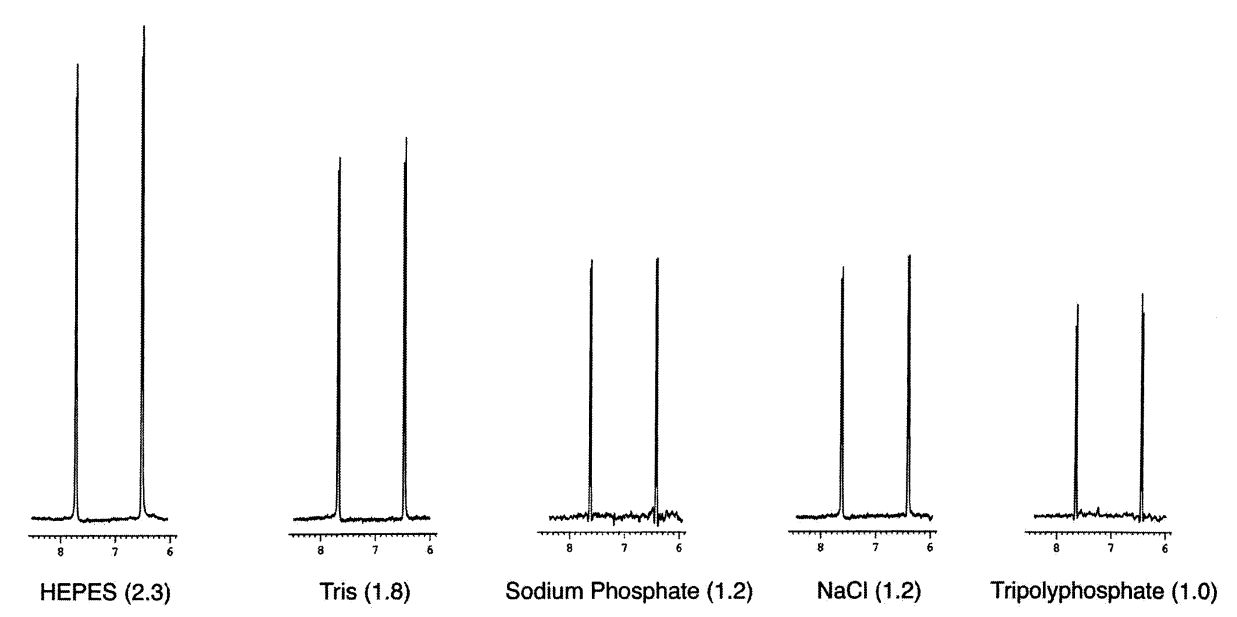
In the figure below, we can see that signal-to-noise ratio (value in parentheses) varies by 2-fold depending on the type of chemicals used to prepare the NMR buffer.
Sensitivities relative to tripolyphosphate of a 2 mM p-aminobenzoic acid sample dissolved in 200 mM buffer. Source: Kelly et al (2002)
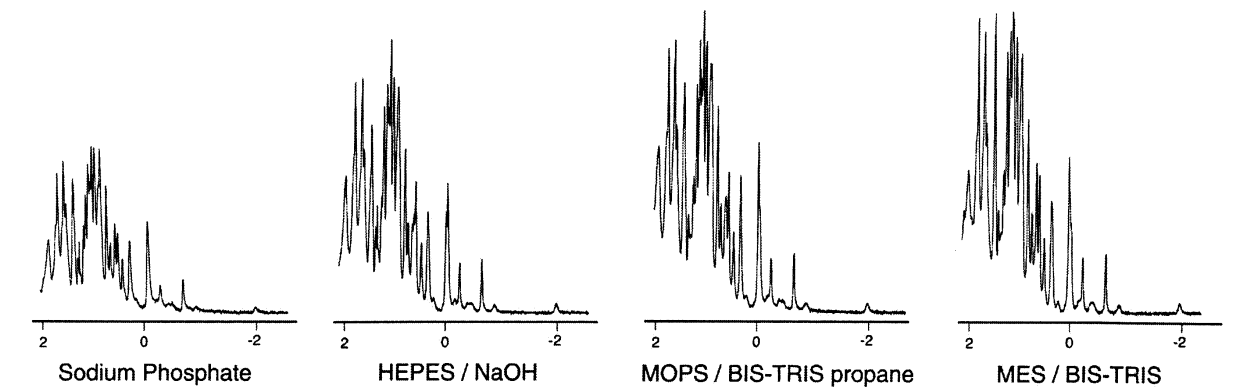
Illustrated below is a similar effect on a biological sample (lysozyme in this case).
Source: Kelly et al (2002)
...
The sample has to be optically clear and free of particles in suspension. A 5-minute centrifugation, or a filtration through a 0.22 µm filter, will remove any suspended matter.
...