| Tabs Page |
|---|
| id | Description |
|---|
| title | Description |
|---|
| Olympus BX63 upright microscopeP-G Desmarais Building, Room 5308-2
Advanced Microscope Tier 1 usage price Instrument awarded to Dr Marina Martinez by the Canadian Foundation for Innovation (CFI)
Simple Microscope usage price
Applications - Transmitted light, Bright-field
- Phase contrast
- Polarized light, DIC
Fluorescence - Color camera
Light sources
| Développer |
|---|
| title | X-Cite NOVEM XT910 complete specifications |
|---|
|
| Source | ID | Filter type | Excitation wavelengths (nm) | Compatible fluorophores | Nominal power (mW) |
|---|
| 365 |
| Violet | 395/25 | Bandpass | [382-407] | DAPI, Hoechst |
| 295| 435 | Blue
| CFP | | Bandpass | [430-450] | CFP | 256 | | Cyan | 470/24 | Bandpass | [458-482] | FITC, GFP | 196 | | Teal | 510/25 | Bandpass | [497-522] | YFP | 62 | | Green/Yellow | 550/15 | Bandpass | [542-557] | TRITC, Cy3 | 260 | Green/Yellow (Storage) | 575/25 | Bandpass | [562-587] | mCherry | 310 | | Red | 640/30 | Bandpass | [625-655] | Cy5 | 231 |
|
Objectives - 20x/0.5 Air Ph1 WD 2.1
- 60x/1.4 Oil DIC WD 0.13
- 100x/1.45 Oil Ph3 WD 0.13
- 100x/1.45 Oil DIC WD 0.13
- 4x/0.2 Air WD 20
- 20x/0.75 Air DIC WD 1.0
| | 475 |
| FITC, GFP |
| | 500-600 | -
[500-600] |
|
| 509/22
[498-520] | YFP |
| 554/23
[542-565] | TRITC |
| 578/21
[567-588] | mCherry |
| | 635 |
| Cy5
|
| 735 |
| Cy7 |
|
|
Objectives 4x/0.16 Air WD 16 10x/0.4 Air WD 3.1 20x/0.75 Air WD 0.6 40x/0.95 Air WD 0.18 60x/1.35 Oil WD 0.15 100x/1.4 Oil WD 0.13 Empty
| Développer |
|---|
| Développer |
|---|
| title | Objectives complete specifications |
|---|
|
| Name | Brand | Full name | Identifier | Working distance | Nom | Marque | Nom complet | Identifiant | Ouverture numérique | Immersion | Distance de travail (mm) | Transmittance
(% [nm]) |
|---|
Techniques | Cover glass thickness | Technique | Épaisseur du couvre-objet (mm) |
|---|
| 1 |
20x5 Ph1Nikon20x5 Plan Fluor Ph1MRH10201 | 2.1 | >80% [400-750] | BF, Pol, PhC, Fluo | 0.17 | 2 | 60x/1.4 Oil DIC | Nikon | 60x/1.4 Oil Plan Apo Lambda DIC N2 | MRD01605 | 0.13 | >80% [475-725]| UPlanSApo | | 0.16 | Air | 16 | >90% at 550nm
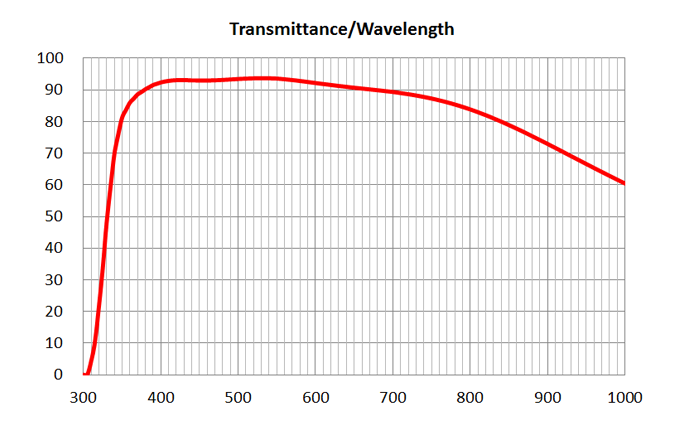 Image Added Image Added | BF, Fluo | - | | 2 | 10x/0.4 Air | Olympus | 10x/0.4 Air
UPlanSApo | UPLXAPO10X.pdf | 0.4 | Air | 3.1 | ~90% at 550nm
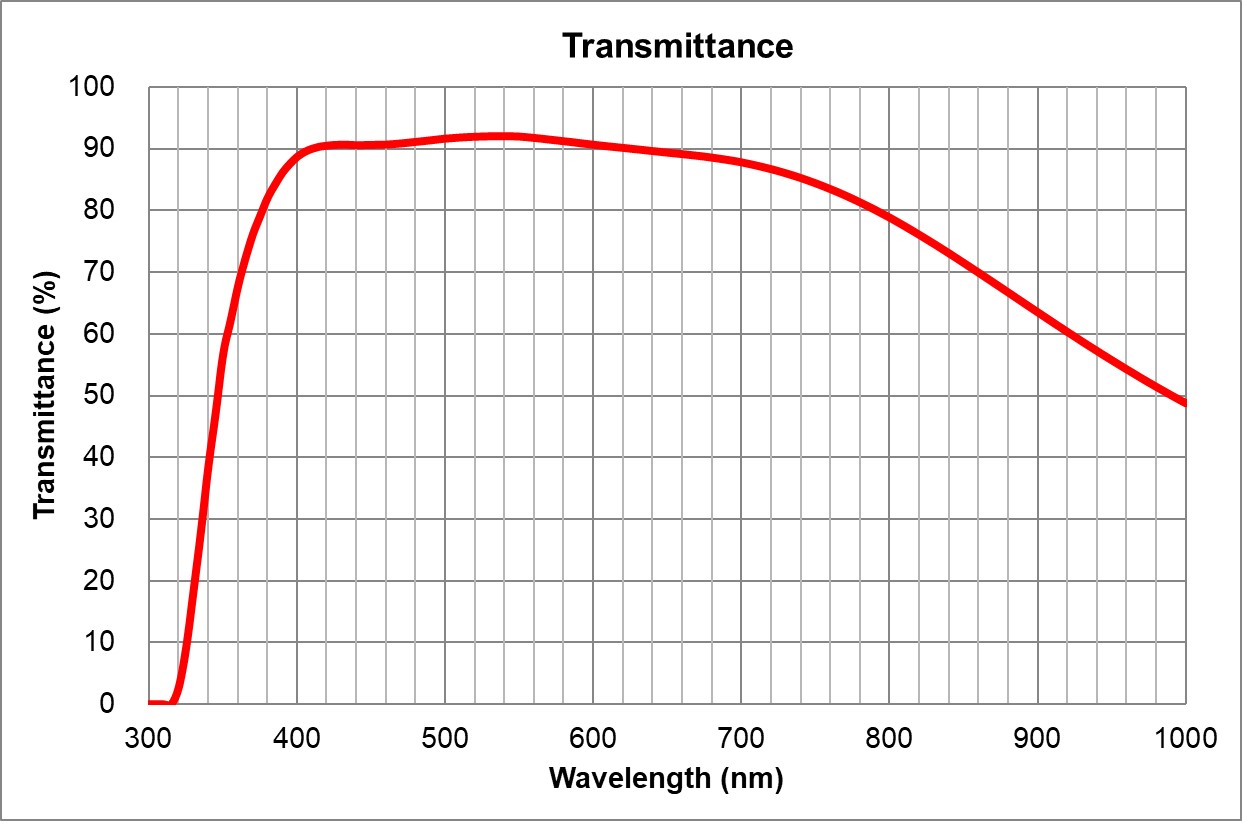 Image Added Image Added | BF, Pol, DIC, Fluo | 0.17 | | 3 |
100x145 Oil Ph3Nikon100x1.45 Oil Plan Apo Lambda Ph3MRD3190513>80% [475-750] | | 75 | | 0.6 | >90% at 550nm
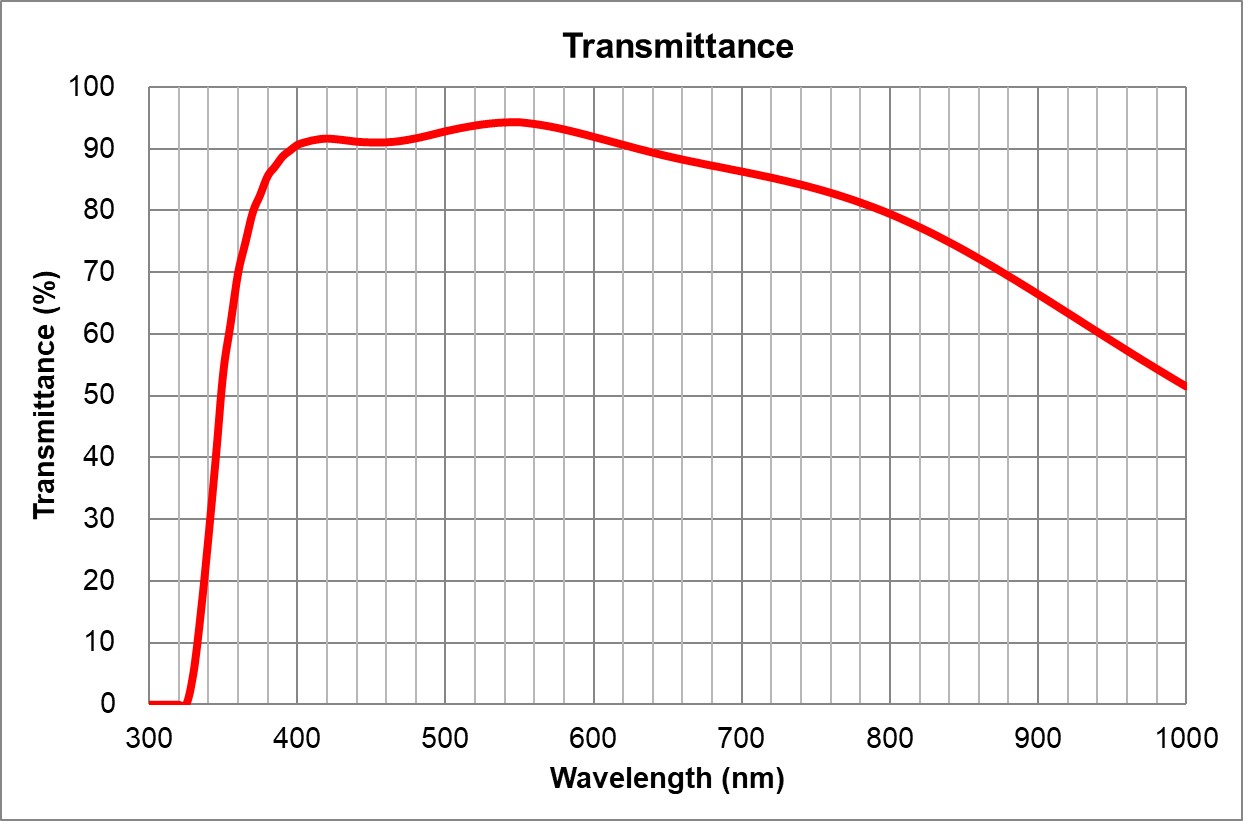 Image Added Image Added | BF |
BF PhC100x1.45 Oil DICNikon | 100x/1.45 Oil Plan Apo Lambda DIC N2 | MRD01905 | 0.13 | >80% [475-750] | BF, Pol, DIC, Fluo | 0.17 | | 5 | 4x/0.2 Air | Nikon | 4x/0.2 Air Plan Apo Lambda | MRD00045 | 20 | >80% [400-1000] | BF, Fluo | 0.17 | 6 | 20x/0.75 Air DIC | Nikon | 20x/0.75 Air Plan Apo Lambda DIC N2 | MRD00205 | 1.0 | 0.95 Air | Olympus | 40x/0.95 Air
UPlanSApo | | 0.95 | Air | 0.18 | ~90% at 550nm
 Image Added Image Added | BF, Pol, DIC, Fluo | Adjustable 0.11-0.23 | | 5 | 60x/1.35 Oil | Olympus | 60x/1.35 Oil
UPlanSApo | | 1.35 | | 0.15 | ~90% at 550nm
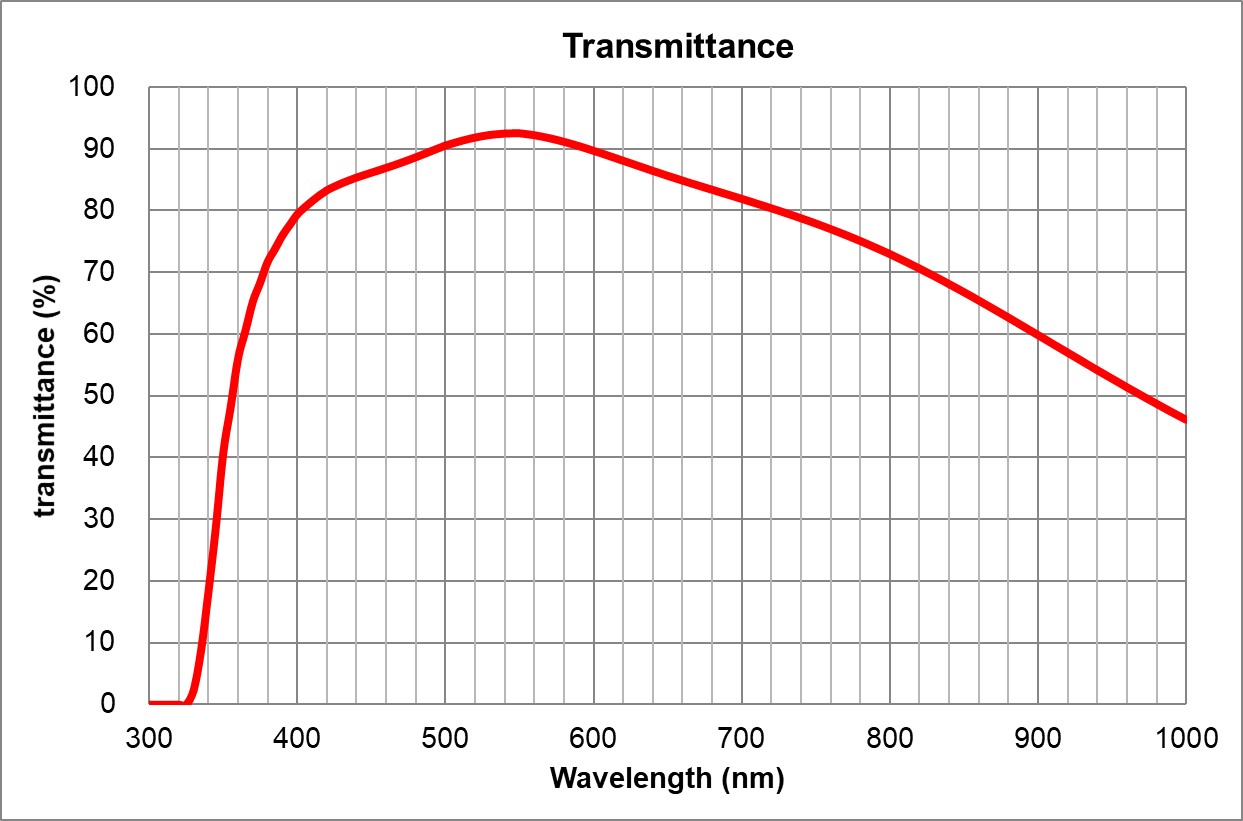 Image Added Image Added |
>80% [400-950] |
Filters DAPI GFP/FITC (CFP) Cy5 DAPI/GFP/Cy3/Cy5 (requires Cy3 filter in Lumencor SpectraX Green/Yellow position) - DIC Analyzer
CFP/YFP/mCherry (requires mCherry filter in Lumencor SpectraX Green/Yellow position)
| Développer |
|---|
| | title | Filters complete specifications |
|---|
| | Position | Name | Brand | ID | Excitation Filter | Dichroic mirror | Emission Filter | Comments |
|---|
| 1 | DAPI | Nikon | DAPI-U HQ | 395/25x
[383-408] | 425LP | 460/50m
[435-485] | C-FL-C DAPI-U HQ | 2 | GFP | Semrock | GFP-4050B-000 | 466/40x
[446-486] | 495LP | 525/50m
[500-550]Nikon ID 96372 | | 3 | Cy5 | Semrock | Cy5-5070A | 617/55x
[590-645] | 652LP | 697/77m
[659-736] | Nikon ID 96376 | | 4 | DAPI/GFP/Cy3/Cy5 | Semrock | C182279 | None | 409/493/573/652 | 432/515/595/681 | 77074160 Custom Quad C182279.
Excitation filters are in the Lumencor SpectraX light source | | 5 | DIC Analyzer | Nikon | Ti2-C-DICACL | Not Applicable | Not Applicable | Not Applicable | Used for DIC imaging | | 6 | CFP\YFP\mCherry | Semrock | C1997767 | None | 459/526/596 | 475/543/702 | Excitation filters are in the Lumencor SpectraX light source | | 6 | | Olympus | 100x/1.4 Oil
UPlanSApo | | 1.4 | | 0.13 | ~90% at 550nm
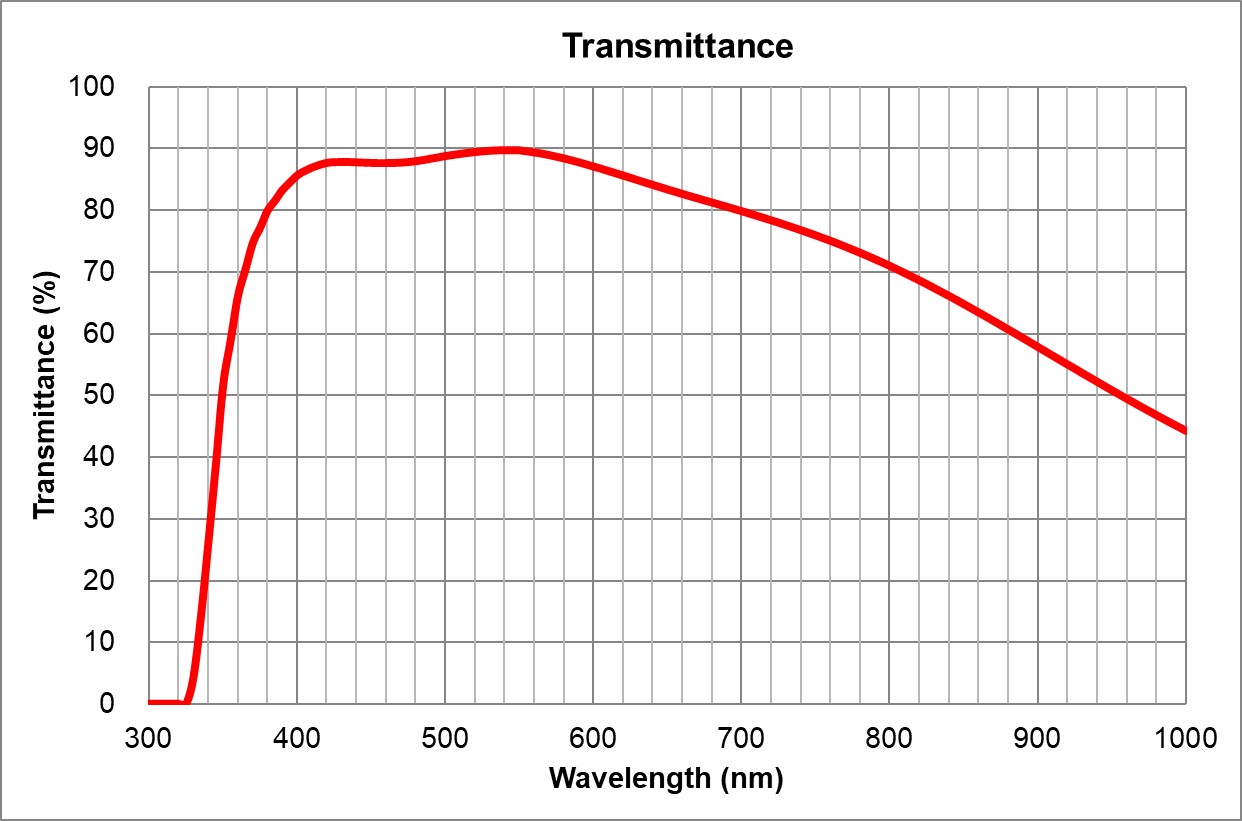 Image Added Image Added | BF, Pol, DIC, Fluo | 0.17 | | 7 | |
|
|
|
| |
|
|
|
|
BF: Bright-field
Pol: Polarized light
DIC: Differential interference contrast
Fluo: Fluorescence |
Filters - Empty
- DAPI
- FITC
- TRITC
- mCherry
- Empty
- Empty
- U-FDICT DIC Analyzer (Nomarski prism)
| Développer |
|---|
| title | Filters complete specifications |
|---|
|
| Position | Name | Brand | ID | Excitation Filter | Dichroic mirror | Emission Filter | Comments |
|---|
| 1 | Empty |
|
|
|
|
|
| | 2 | DAPI | Olympus | U-FUNA | 360-370 | 410LP | 420-460 |  Image Added Image Added
| | 3 | FITC | Olympus | U-FBNA | 470-495 | 505LP | 510-550 |  Image Added Image Added
| | 4 | TRITC | Olympus | U-FGWA | 530-550 | 570LP | 575-625 |  Image Added Image Added
| | 5 | mCherry | Olympus | U-FYW | 540-585 | 595LP | 600LP |  Image Added Image Added
| | 6 | Empty |
|
|
|
|
|
| | 7 | Empty |
|
|
|
|
|
| | 8 | DIC Analyzer | Olympus | U-FDICT DIC | - | - | - | DIC Analyzer |
|
Detector Olympus DP80 Dual sensors (color and monochrome) CCD camera, 6.45 x 6.45 um pixel size,15 images/s at full frame 55% QE at 550nm
Color mode 4080 x 3072 pixels, 24-bit
Monochrome mode 1360 x 1024 pixels, 14-bit
Olympus_DP80 Camera_User Guide.pdf
| Tabs Page |
|---|
| id | User Guide |
|---|
| title | User Guide |
|---|
|
| | Tabs Page |
|---|
| id | User Guide |
|---|
| title | User Guide |
|---|
| | UI Expand |
|---|
| - Turn on the computer (#1)
- Remove the cover from the microscope
- Turn on the microscope power bar computer (#2#1)
If incubation is required, turn - Turn on the
Okolab incubation module (#3A), the Lauda water bath (#3B) and open the CO2 (#3C) and N2 (#3D) tanks| Avertissement |
|---|
Make sure the humidifier and the water bath are clean and properly filled with distilled water. |
- microscope power bar(#2)
Turn on the microscope touchscreen (switch is located at the back on the right of the touch screen) (#3) Use your UdeM Use your UdeM credentials to log in to Windows Start the software NIS-Elements| Info |
|---|
The first time you use the instrument, you need to import the microscope settings into the software. To do this follow the instructions in the First use |
protocol
- Start the software CellSens Dimension
|
| UI Expand |
|---|
| When using the microscope for the first time, you need to import the microscope settings into the software. You will usually do this during the training session.
This procedure can also be performed if something is not working properly and if you want to reset the software to its original settings. | Remarque |
|---|
This process will delete all experiment protocols and reset the software to the original settings for this specific microscope. |
- If open, close the software CellSens Dimension and wait until it is completely closed (up to 30 seconds)
- On your Desktop open the folder Documentation
- Double-click Settings for BX-63
- A script will run and a black window will briefly appear
- You can now reopen the software CellSens Dimension
Save your dataClose NIS-Elements software
Transfer your data to the D: drive (Data Storage) or to your external drive and delete it from the local C: driveTurn off the computerIf oil objectives were used, clean it with lens cleaner and lens paper (not Kimwipes)If incubation was used, turn off the Okolab incubation module (#3A),Lauda water bath (#3B) and close the CO2 (#3C) and N2 (#3D) tanksWait until the SpectaX has cooled down and turn off the microscope power bar (#2)Cover the microscope| Remarque |
|---|
| | Collect your samples, especially those in the microscopeLeave the microscope and workspace clean
| UI Expand |
|---|
| - Files can be saved temporarily (during acquisition) to local C: drive (desktop)
- At the end of each session, copy your data to your external drive and delete it from local C: drive
- You can store your files on the D: drive (Data Storage). If you do, please create one folder per laboratory using the principal investigator's last name. Inside, create a folder per user using the following nomenclature (First Name_Last Name).
| Remarque |
|---|
| In any case, do not store your files on the C: drive. |
| UI Expand |
|---|
| | Ancre |
|---|
FirstUse | FirstUse | When using the microscope for the first time, you need to import the microscope settings into the software. You will usually do this during the training session.
This procedure can also be performed if something is not working properly and if you want to reset the software to its original settings.| Remarque |
|---|
This process will delete all experiment protocols and reset the software to the original settings for this specific microscope. |
- If open, close NIS-Elements and wait until it is completely closed (up to 30 seconds)
- On your Desktop open the Softwares folder
- Open NIS Settings Utility
- Click on the Import tab
- Click on Browse
- Navigate to your Desktop
- Select the file Nikon-Ti2 Settings.bin
- Click Select
- Select all items
- Click Import
- Click OK
- Close the NIS Settings
- You can now reopen NIS-Elements
| | Tabs Page |
|---|
| id | Light path |
|---|
| title | Light path |
|---|
| The following schematics depict the light path for transmitted (bright-field and Phase Contrast) and reflected (fluorescence) lights.| PDF |
|---|
| name | LightPath_Nikon Ti2.pdf |
|---|
|
| Tabs Page |
|---|
| Available manuals| Tabs Page |
|---|
| | UI Expand |
|---|
| - Check condenser filters if labels match
- Check why 20x/0.75 DIC objective has no DIC prism
- Condenser DIC prism is DIC N1 while objective requires N2
- Check why at 4x Condenser gives a black shadow on the righ side of the image
- Adjust Objective XYZ offset
- Check plane because FOV is not flat
|
| UI Expand |
|---|
| expanded | true |
|---|
| title | 2023-11-29 |
|---|
| - Fixed condenser holder loose
- 20x/0.5 Ph1 dirty or damaged likely dirty with varnished
|
| UI Expand |
|---|
| - Added network cable between Acquisition and Processing workstations
- Added shortcuts in Desktop\Documentation
|
| UI Expand |
|---|
| - Clean 60x objective (nail polish on it !)
- Fix damaged stage insert
- Stage insert tilt adjustment
- Objective XY offset adjustments
|
| UI Expand |
|---|
| - Installed new objective 4x/0.2 Plan Apo Lambda
- Updated configuration file
|
| UI Expand |
|---|
| - Computer maintenance
- Imaging test slides
- Re-install DCAM API drivers v22.2.6391
- Okolab incubation complete maintenance
- Disassemble Okolab stage insert
- Disconnect the pipes
- Disconnect the Lauda water-bath
- Clean everything with 10% acetic acid (excepted the plastic pipes and washers)
- Rinse with regular tap water 2 times
- Rinse with distilled water once
- Dry, reassemble and reconnect
- Refill with the Lauda water-bath with 3L of milliQ water
- Set the Okolab temperature Control Mode to Chamber
- Set the Okolab temperature to 65°C
- Let run for 1h. This will sterilize the water.
- Check for any leakage
- Set the Okolab temperature Control Mode back to Sample
- Set the Okolab temperature back to 37°C
|
| UI Expand |
|---|
| - Cleaning objectives
- Control for incubation chamber liquid intrusion
|
| UI Expand |
|---|
| - CO2 Calibration for Okolab: Offset of 1%
|
| UI Expand |
|---|
| - 20x/0.75 DIC objective was dirty and has been cleaned
- Printed and displayed a Memo about available air and oil objectives
- Adjustment of camera angle
- Objective calibration
- Objective XY offset
- Updated NIS settings
- Added light path schematics to wikipedia
|
| UI Expand |
|---|
| - Added label on gas bottles
- Added line mark on humidifier
|
| UI Expand |
|---|
| - 512 GB SSD installed for OS
- Windows 10 Installation
- BIOS updated to v2.47
|
| Tabs Page |
|---|
| id | Technical Datasheet |
|---|
| title | Technical Datasheet |
|---|
| Stand- Nikon Ti2-E inverted Serial 540156 System 170110-Sys-006287
Light sources- Transmitted LED light
- ND32 filter
- IR filter
- Manual Polarizer
- Lumencor SpectraX 6-NII-SE Serial 9409
CondenserObjectives- 20x/0.5 Air Ph1 WD 2.1
- 60x/1.4 Oil DIC WD 0.13
- 100x/1.45 Oil Ph3 WD 0.13
- 100x/1.45 Oil DIC WD 0.13
- 4x/0.2 Air WD 20
- 20x/0.75 Air DIC WD 1.0
Stage- Motorized stage Ti2 SHU compatible Serial 127808
- Remote control joystick Ti2-S-JS Serial 127976
- Inserts
- Multi-well plate Ti2-S-HW with tilt adjustment (no incubation)
- Combo slide 3cm dish Ti2-S-HU with tilt adjustment (no incubation)
- Okolab H101-CellASIC Frame with perfusion ports
- 1 x 35mm petri dish 1x35-M + cover
- 2 Chamber Slide 2xGS-M+ cover
- 1 Multi-well for oil objectives MW-OIL
- 6-well plate 6MW+ cover
- 1 CellASIC + cover
Filters - DAPI Cube Ex 383-408 DAPI-U DM 425 BA 435-485
- GFP Cube Semrock 96372 M349727 17
- Cy5 Cube Semrock 96376 M351081 8
- 77074160 Custom Quad C182279 Polychroic and quad bandpass emitter for use with the following single bandpass filters: ET395/25x, ET470/24x, ET550/15x, ET640/30x
- DIC Analyser Ti2-C-DICACL
- C197767 7707\4656 CFP\YFP\mCherry XT
Detector- Hamamatsu ORCA Flash V2 C11440-22CU CMOS Monochrome Camera 2048 x 2048 pixels, 16-bit, 30fps at full resolution Serial 101081
Workstation- HP Z440 Workstation
- Intel Xeon E5-1620 v4 @ 3.5GHz
- RAM 32 GB DDR4 2400 MHz ECC (4 x 8 GB)
- OS 500 GB SSD 530 MB/s
- 4 TB HD Data Storage (2 x 2 TB spanned volume) 130 MB/s
- Video Card nVidia GTX 1080 8GB DDR5 dedicated memory
- Monitor HP Z24i display 24' 1920x1200
- Software NIS-Elements AR v5.02
Incubation- Okolab BoldLine Temperature unit Serial 284-1058 H101 T Unit BL
- Okolab BoldLine CO2/O2 Unit 0-10/1-18 Serial 088-1102
- Okolab OkoTouch Serial 118-224
- Lauda water-bath Model Eco RE415 S LCK 4910 Serial LCK-4910-16-0006 Okolab 1322-1006 2017-05-30
Consumables- CO2 Tank
- N2 Tank
- Liquid Light Guide
|
| Tabs Page |
|---|
|









